【第64期】前沿靶點(diǎn)速遞:每周醫(yī)學(xué)研究精選
日期:2025-11-18 16:30:47
01、靶點(diǎn):SKP2、STUB1
應(yīng)用:腫瘤免疫治療
來(lái)源:A rapid imaging-based screen for induced-proximity degraders identifies a potent degrader of oncoprotein SKP2.Nat Biotechnol,2025 Sep 10

02、靶點(diǎn):Munc13-4
應(yīng)用:腫瘤免疫治療
來(lái)源:Munc13-4 mediates tumor immune evasion by regulating the sorting and secretion of PD-L1 via exosomes.Nat Commun,2025 Oct 13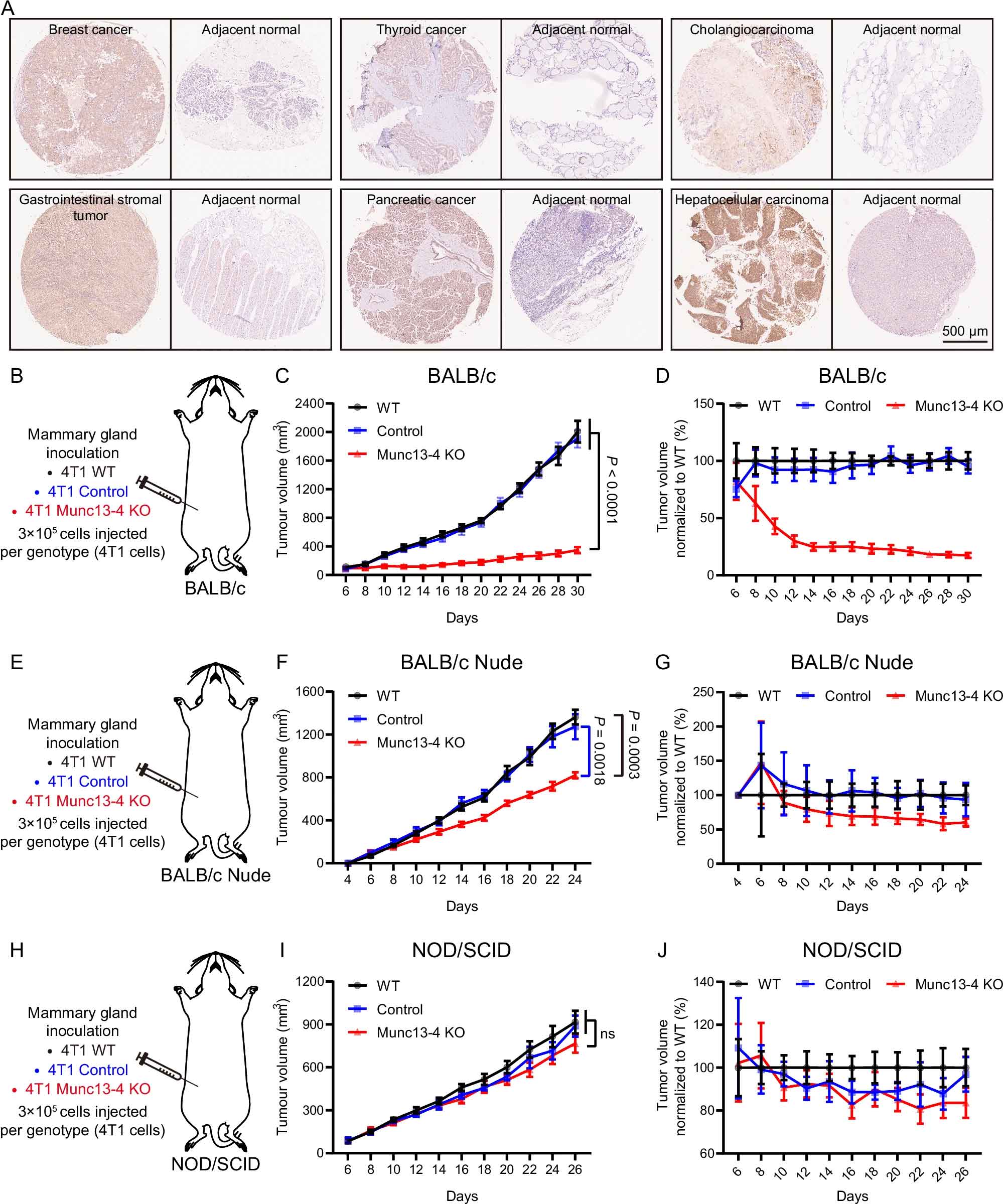
2025年10月13日,華中科技大學(xué)馬聰教授團(tuán)隊(duì)聯(lián)合中國(guó)科學(xué)院廣州生物醫(yī)藥與健康研究院何俊教授團(tuán)隊(duì)在《Nature Communications》發(fā)表研究論文,揭示了Munc13-4通過(guò)調(diào)控外泌體PD-L1的分選與分泌促進(jìn)腫瘤免疫逃逸的新機(jī)制。研究發(fā)現(xiàn),Munc13-4在多種腫瘤組織中表達(dá)上調(diào),其缺失可增強(qiáng)T細(xì)胞介導(dǎo)的抗腫瘤反應(yīng)。Munc13-4通過(guò)與PD-L1結(jié)合并招募HRS形成三元復(fù)合物,調(diào)控PD-L1的分選,并協(xié)同Rab27a介導(dǎo)多囊泡體錨定至細(xì)胞膜,促進(jìn)外泌體分泌。干擾素γ通過(guò)調(diào)控Munc13-4與HRS的修飾動(dòng)態(tài)控制PD-L1分選。基于此,團(tuán)隊(duì)開(kāi)發(fā)了靶向Munc13-4與PD-L1互作的小肽P-pep,可阻斷PD-L1進(jìn)入外泌體,顯著增強(qiáng)腫瘤微環(huán)境中T細(xì)胞的浸潤(rùn)與活化,抑制腫瘤生長(zhǎng),為免疫治療提供了新策略。
03、靶點(diǎn):BRRF2
應(yīng)用:EBV相關(guān)腫瘤免疫治療
來(lái)源:Extracellular vesicles derived EBV tegument protein BRRF2 suppresses cGAS phase separation to promote anti-viral innate immune evasion.Nat Commun,2025 Oct 10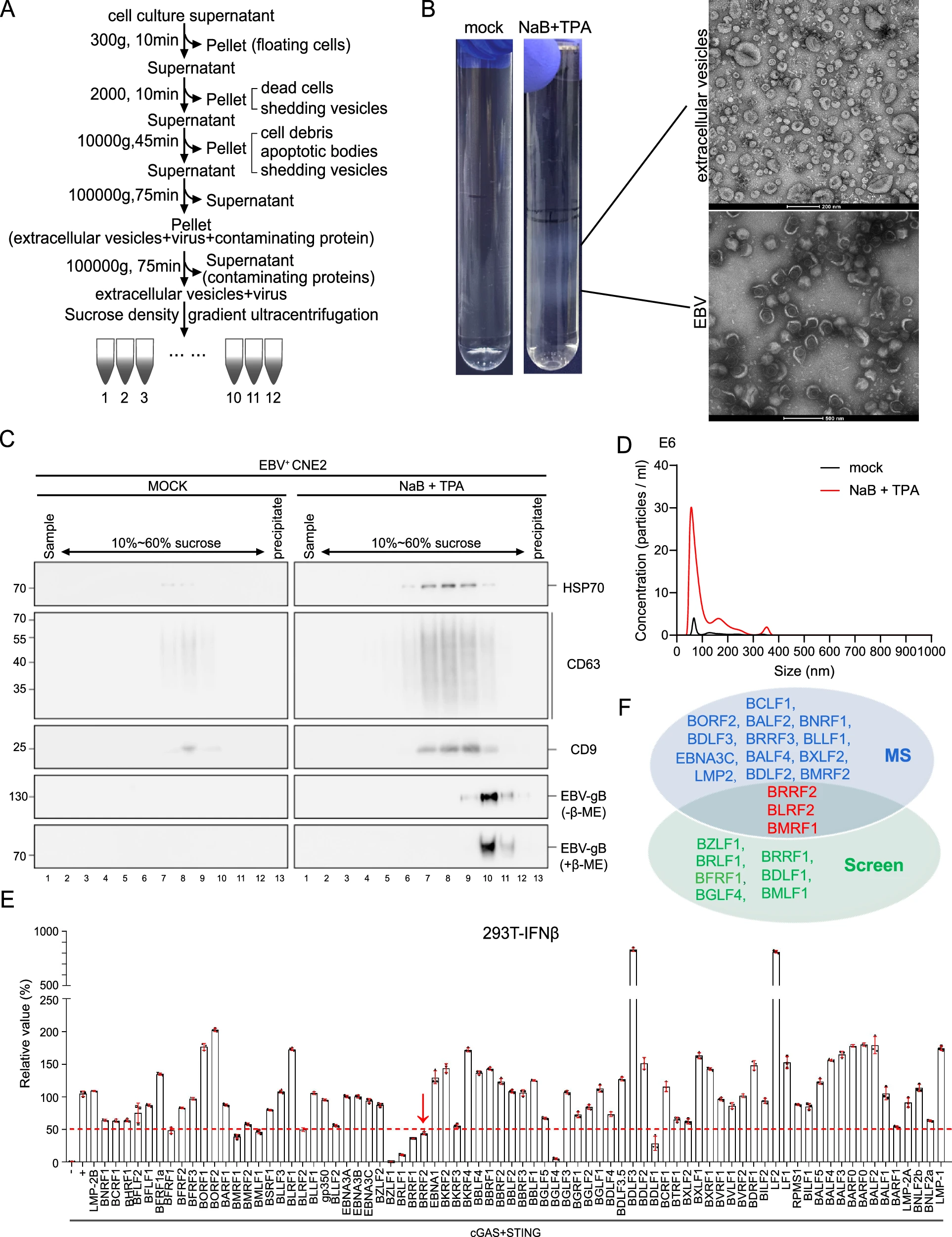
04、靶點(diǎn):RAB22A
應(yīng)用:結(jié)直腸癌的治療
來(lái)源:RAB22A triggers intercellular chemoresistance transmission in colorectal cancer by promoting exosome release via the PKM2-pSNAP23 axis.Oncogene,2025 Nov
05、靶點(diǎn):USP2
應(yīng)用:MASLD的治療
來(lái)源:USP2 promotes metabolic dysfunction-associated steatotic liver disease progression via stabilization of PPARγ.Cell Death Differ,2025 Sep 24
06、靶點(diǎn):PFKL
應(yīng)用:解決實(shí)體瘤耐藥問(wèn)題
來(lái)源:The noncanonical function of liver-type
phosphofructokinase potentiates the efficacy of HDAC inhibitors in cancer.Signal Transduct Target Ther,2025 Oct 14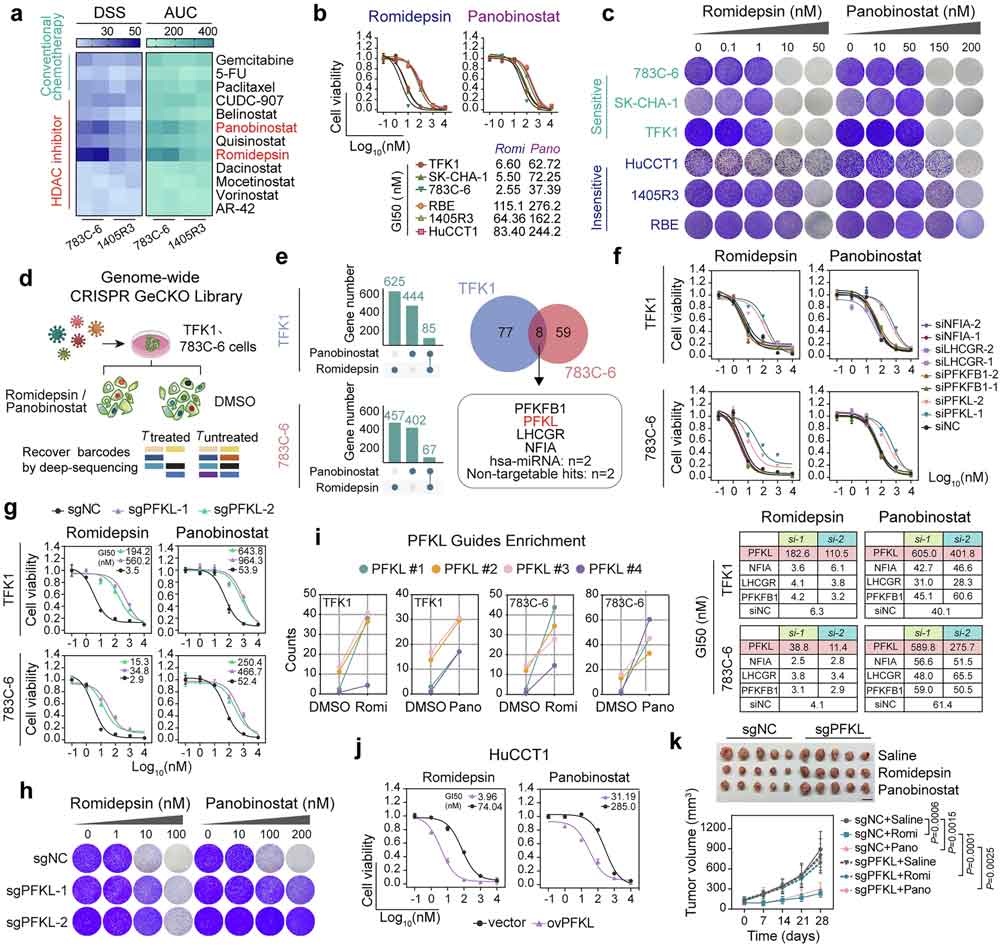
推薦產(chǎn)品
參考文獻(xiàn)
[1]A rapid imaging-based screen for induced-proximity degraders identifies a potent degrader of oncoprotein SKP2.Nat Biotechnol,2025 Sep 10
[2]Munc13-4 mediates tumor immune evasion by regulating the sorting and secretion of PD-L1 via exosomes.Nat Commun,2025 Oct 13
[3]Extracellular vesicles derived EBV tegument protein BRRF2 suppresses cGAS phase separation to promote anti-viral innate immune evasion.Nat Commun,2025 Oct 10
[4]RAB22A triggers intercellular chemoresistance transmission in colorectal cancer by promoting exosome release via the PKM2-pSNAP23 axis.Oncogene,2025 Nov
[5]USP2 promotes metabolic dysfunction-associated steatotic liver disease progression via stabilization of PPARγ.Cell Death Differ,2025 Sep 24
[6]The noncanonical function of liver-type phosphofructokinase potentiates the efficacy of HDAC inhibitors in cancer.Signal Transduct Target Ther,2025 Oct 14
*免責(zé)聲明:華美生物內(nèi)容團(tuán)隊(duì)僅是分享和解讀公開(kāi)研究論文及其發(fā)現(xiàn),本文僅作信息交流,文中觀點(diǎn)不代表華美生物立場(chǎng),請(qǐng)理解。
應(yīng)用:腫瘤免疫治療
來(lái)源:A rapid imaging-based screen for induced-proximity degraders identifies a potent degrader of oncoprotein SKP2.Nat Biotechnol,2025 Sep 10

圖源:10.1038/s41587-025-02793-8[1]
2025年9月10日,廈門(mén)大學(xué)鄧賢明團(tuán)隊(duì)與中國(guó)科學(xué)院分子細(xì)胞科學(xué)卓越創(chuàng)新中心姜海團(tuán)隊(duì)在《Nature Biotechnology》發(fā)表研究論文,構(gòu)建了基于“死亡-存活”表型轉(zhuǎn)換的高通量蛋白降解劑篩選體系DEFUSE,并發(fā)現(xiàn)新型分子膠降解劑SKPer1。SKPer1通過(guò)泛素連接酶STUB1高效降解腫瘤驅(qū)動(dòng)蛋白SKP2,展現(xiàn)出顯著抗腫瘤活性。該研究突破了“不可成藥”靶點(diǎn)SKP2的治療瓶頸,為分子膠降解劑的系統(tǒng)性篩選提供了全新技術(shù)路徑,還拓展了分子膠技術(shù)體系,具有重要意義。02、靶點(diǎn):Munc13-4
應(yīng)用:腫瘤免疫治療
來(lái)源:Munc13-4 mediates tumor immune evasion by regulating the sorting and secretion of PD-L1 via exosomes.Nat Commun,2025 Oct 13

圖源:10.1038/s41467-025-64149-9[2]
2025年10月13日,華中科技大學(xué)馬聰教授團(tuán)隊(duì)聯(lián)合中國(guó)科學(xué)院廣州生物醫(yī)藥與健康研究院何俊教授團(tuán)隊(duì)在《Nature Communications》發(fā)表研究論文,揭示了Munc13-4通過(guò)調(diào)控外泌體PD-L1的分選與分泌促進(jìn)腫瘤免疫逃逸的新機(jī)制。研究發(fā)現(xiàn),Munc13-4在多種腫瘤組織中表達(dá)上調(diào),其缺失可增強(qiáng)T細(xì)胞介導(dǎo)的抗腫瘤反應(yīng)。Munc13-4通過(guò)與PD-L1結(jié)合并招募HRS形成三元復(fù)合物,調(diào)控PD-L1的分選,并協(xié)同Rab27a介導(dǎo)多囊泡體錨定至細(xì)胞膜,促進(jìn)外泌體分泌。干擾素γ通過(guò)調(diào)控Munc13-4與HRS的修飾動(dòng)態(tài)控制PD-L1分選。基于此,團(tuán)隊(duì)開(kāi)發(fā)了靶向Munc13-4與PD-L1互作的小肽P-pep,可阻斷PD-L1進(jìn)入外泌體,顯著增強(qiáng)腫瘤微環(huán)境中T細(xì)胞的浸潤(rùn)與活化,抑制腫瘤生長(zhǎng),為免疫治療提供了新策略。
03、靶點(diǎn):BRRF2
應(yīng)用:EBV相關(guān)腫瘤免疫治療
來(lái)源:Extracellular vesicles derived EBV tegument protein BRRF2 suppresses cGAS phase separation to promote anti-viral innate immune evasion.Nat Commun,2025 Oct 10

圖源:10.1038/s41467-025-64037-2[3]
2025年10月10日,中山大學(xué)腫瘤防治中心鐘茜、曾木圣研究團(tuán)隊(duì)在《Nature Communications》發(fā)表研究,揭示EB病毒(EBV)通過(guò)細(xì)胞外囊泡(EVs)遞送BRRF2蛋白,破壞宿主天然免疫防線,助力腫瘤免疫逃逸。EBV是與多種癌癥相關(guān)的人類致癌病毒,其在鼻咽癌等腫瘤中通過(guò)EVs將BRRF2精準(zhǔn)遞送至巨噬細(xì)胞。BRRF2通過(guò)與cGAS蛋白特異性結(jié)合,阻斷DNA識(shí)別、瓦解相分離、抑制酶活性,癱瘓cGAS-STING信號(hào)通路,抑制抗病毒免疫應(yīng)答。此外,BRRF2還促進(jìn)EBV復(fù)制,形成“免疫逃逸-病毒復(fù)制”的正反饋環(huán)路。該研究為克服EBV相關(guān)腫瘤免疫治療抵抗提供了新方向。04、靶點(diǎn):RAB22A
應(yīng)用:結(jié)直腸癌的治療
來(lái)源:RAB22A triggers intercellular chemoresistance transmission in colorectal cancer by promoting exosome release via the PKM2-pSNAP23 axis.Oncogene,2025 Nov

圖源:10.1038/s41388-025-03566-y[4]
江南大學(xué)附屬醫(yī)院腫瘤研究所黃朝暉教授/殷媛研究員團(tuán)隊(duì)在《Oncogene》發(fā)表研究,揭示外泌體介導(dǎo)結(jié)直腸癌化療耐藥的新機(jī)制。研究發(fā)現(xiàn),小G蛋白R(shí)AB22A不僅直接促進(jìn)結(jié)直腸癌細(xì)胞化療耐藥,還通過(guò)促進(jìn)富含RAB22A-PKM2的外泌體分泌,誘導(dǎo)腫瘤微環(huán)境中其他癌細(xì)胞獲得性耐藥。RAB22A通過(guò)抑制PKM2的泛素化和降解,促進(jìn)SNAP-23的磷酸化,增加外泌體分泌。耐藥細(xì)胞分泌的外泌體攜帶RAB22A和穩(wěn)定PKM2,被敏感細(xì)胞吞噬后,增強(qiáng)其耐藥能力。該研究為結(jié)直腸癌化療耐藥的精準(zhǔn)預(yù)測(cè)和靶向逆轉(zhuǎn)提供了新策略。05、靶點(diǎn):USP2
應(yīng)用:MASLD的治療
來(lái)源:USP2 promotes metabolic dysfunction-associated steatotic liver disease progression via stabilization of PPARγ.Cell Death Differ,2025 Sep 24

圖源:10.1038/s41418-025-01589-2[5]
上海交通大學(xué)醫(yī)學(xué)院吳英理課題組在《Cell Death & Differentiation》發(fā)表研究,揭示去泛素化酶USP2通過(guò)穩(wěn)定PPARγ促進(jìn)代謝功能障礙相關(guān)脂肪性肝病(MASLD)的進(jìn)展。研究發(fā)現(xiàn),USP2在MASLD肝臟組織中表達(dá)上調(diào),其特異性去除PPARγ DNA結(jié)合域中第161位賴氨酸上的K48連接的多聚泛素鏈,阻止PPARγ被蛋白酶體降解,增強(qiáng)其穩(wěn)定性和轉(zhuǎn)錄活性,促進(jìn)脂質(zhì)合成相關(guān)基因表達(dá),導(dǎo)致肝細(xì)胞脂質(zhì)蓄積。團(tuán)隊(duì)利用肝靶向GalNAc-siRNA技術(shù)驗(yàn)證了USP2作為治療靶點(diǎn)的潛力,為MASLD的治療提供了新策略。06、靶點(diǎn):PFKL
應(yīng)用:解決實(shí)體瘤耐藥問(wèn)題
來(lái)源:The noncanonical function of liver-type
phosphofructokinase potentiates the efficacy of HDAC inhibitors in cancer.Signal Transduct Target Ther,2025 Oct 14

圖源:10.1038/s41392-025-02443-0[6]
10月14日,海軍軍醫(yī)大學(xué)王紅陽(yáng)院士/董立巍研究員團(tuán)隊(duì)在《Signal Transduction and Targeted Therapy》(IF=52.7)發(fā)表成果,揭示肝型磷酸果糖激酶(PFKL)是提升組蛋白去乙酰化酶抑制劑(HDACi)實(shí)體瘤療效的關(guān)鍵因子。研究以膽管癌為模型,發(fā)現(xiàn)PFKL在細(xì)胞核中調(diào)控HDACi療效和細(xì)胞內(nèi)表觀遺傳動(dòng)力學(xué)。基于此,團(tuán)隊(duì)開(kāi)發(fā)了一種細(xì)胞穿透性治療肽,利用PFKL增強(qiáng)藥物精準(zhǔn)性,顯著提升抗腫瘤效果,為解決實(shí)體瘤耐藥問(wèn)題提供新策略。推薦產(chǎn)品
| 靶點(diǎn) | 重組蛋白 | 貨號(hào) |
| BRRF2 | Recombinant Epstein-Barr virus Tegument protein BRRF2 (BRRF2), partial | CSB-MP365877EFA |
| PFKL | Recombinant Human ATP-dependent 6-phosphofructokinase, liver type (PFKL) | CSB-MP017821HU |
| RAB22A | Recombinant Human Ras-related protein Rab-22A (RAB22A) | CSB-MP891957HU |
| SKP2 | Recombinant Human S-phase kinase-associated protein 2 (SKP2) | CSB-EP613392HU |
| STUB1 | Recombinant Human E3 ubiquitin-protein ligase CHIP (STUB1) | CSB-EP892480HUe1 |
| UNC13D | Recombinant Human Protein unc-13 homolog D (UNC13D), partial | CSB-MP758222HU |
| USP2 | Recombinant Human Ubiquitin carboxyl-terminal hydrolase 2 (USP2) | CSB-MP025710HU |
參考文獻(xiàn)
[1]A rapid imaging-based screen for induced-proximity degraders identifies a potent degrader of oncoprotein SKP2.Nat Biotechnol,2025 Sep 10
[2]Munc13-4 mediates tumor immune evasion by regulating the sorting and secretion of PD-L1 via exosomes.Nat Commun,2025 Oct 13
[3]Extracellular vesicles derived EBV tegument protein BRRF2 suppresses cGAS phase separation to promote anti-viral innate immune evasion.Nat Commun,2025 Oct 10
[4]RAB22A triggers intercellular chemoresistance transmission in colorectal cancer by promoting exosome release via the PKM2-pSNAP23 axis.Oncogene,2025 Nov
[5]USP2 promotes metabolic dysfunction-associated steatotic liver disease progression via stabilization of PPARγ.Cell Death Differ,2025 Sep 24
[6]The noncanonical function of liver-type phosphofructokinase potentiates the efficacy of HDAC inhibitors in cancer.Signal Transduct Target Ther,2025 Oct 14
*免責(zé)聲明:華美生物內(nèi)容團(tuán)隊(duì)僅是分享和解讀公開(kāi)研究論文及其發(fā)現(xiàn),本文僅作信息交流,文中觀點(diǎn)不代表華美生物立場(chǎng),請(qǐng)理解。













