【第74期】前沿靶點(diǎn)速遞:每周醫(yī)學(xué)研究精選
日期:2026-02-04 17:03:22
01、靶點(diǎn):METTL14
應(yīng)用:PDAC疼痛抑郁共病的潛在治療靶點(diǎn)
來源:METTL14 integrates tumor-derived SAM to drive parabrachial epigenetic rewiring in pancreatic cancer.Neuron,2026 Jan 21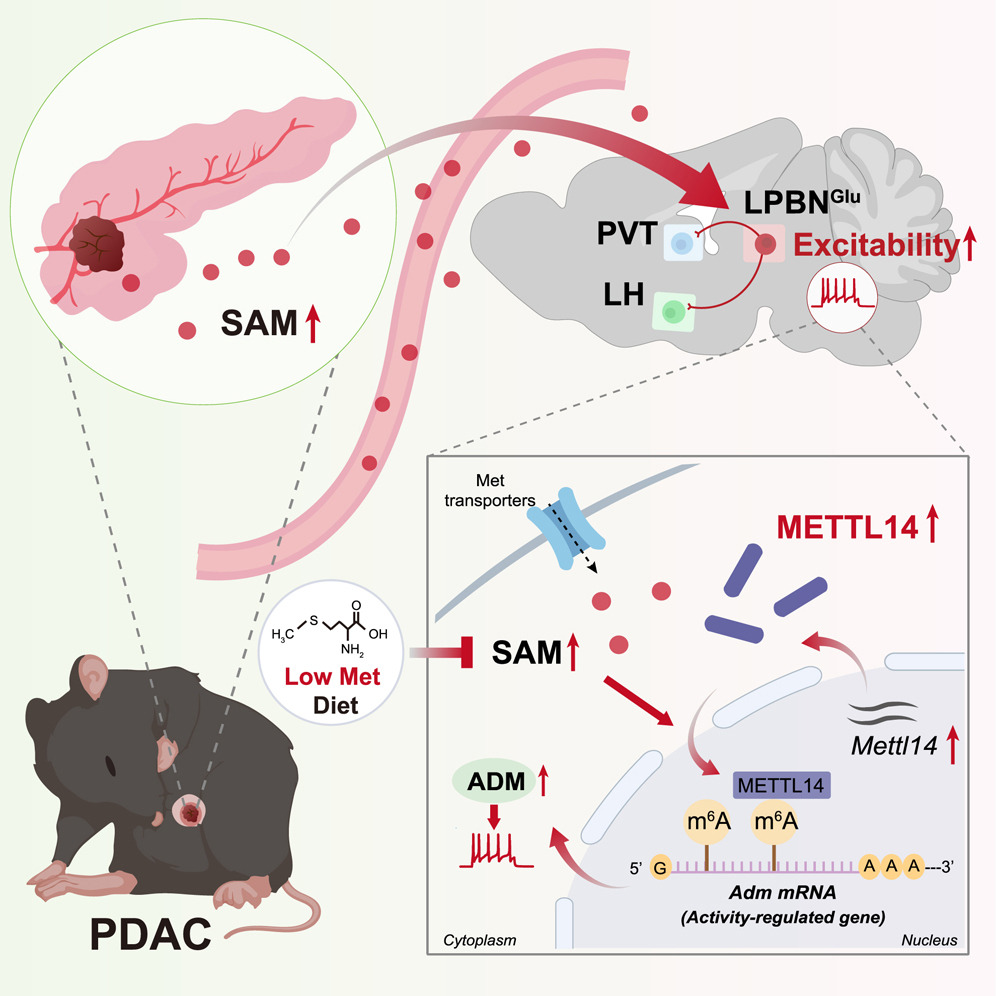
中山大學(xué)謝敬敦團(tuán)隊(duì)11月于《Neuron》報(bào)道,PDAC小鼠血漿及外側(cè)臂旁核(LPBN)內(nèi)腫瘤源S-腺苷甲硫氨酸(SAM)升高,激活谷氨酸能神經(jīng)元METTL14,經(jīng)m6A修飾增強(qiáng)ADM mRNA穩(wěn)定性,使神經(jīng)元超興奮,并通過LPBNGlu→PVTGlu/LHGlu環(huán)路誘發(fā)痛覺過敏與抑郁。敲低METTL14、抑制SAM合成或低甲硫氨酸飲食均可逆轉(zhuǎn)共病并抑制腫瘤,揭示“腫瘤-代謝物-神經(jīng)元”信號(hào)軸,為PDAC疼痛抑郁共病提供可轉(zhuǎn)化干預(yù)策略。
02、靶點(diǎn):AAK1
應(yīng)用:乳腺癌等實(shí)體瘤的潛在治療靶點(diǎn)
來源:AAK1 activation-mediated iron trafficking drives ferroptotic cell death.Nat Commun,2025 Dec 17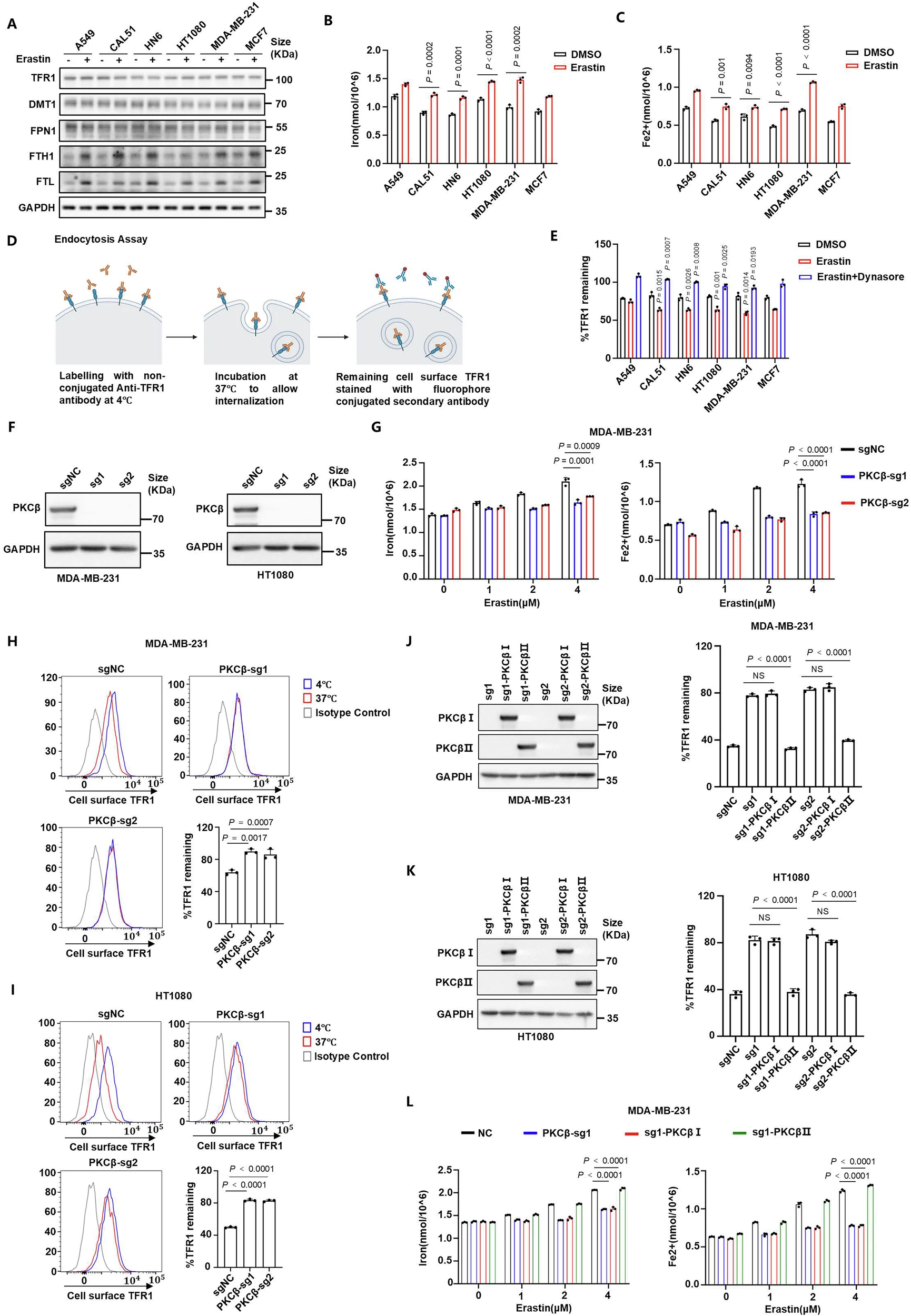
中山大學(xué)朱孝峰團(tuán)隊(duì)12月在Nat Commun報(bào)道,鐵死亡啟動(dòng)時(shí)PKCβII感應(yīng)脂質(zhì)過氧化,并在S670/T674位點(diǎn)磷酸化激活A(yù)AK1,后者磷酸化AP2M1促進(jìn)網(wǎng)格蛋白依賴的轉(zhuǎn)鐵蛋白受體TFR1內(nèi)吞,使胞外鐵大量流入,為脂質(zhì)過氧化物生成提供催化鐵,從而驅(qū)動(dòng)腫瘤細(xì)胞鐵死亡。敲除或抑制PKCβⅡ-AAK1-AP2M1軸均降低鐵攝取、抑制鐵死亡并削弱放療敏感性;激活該軸則增強(qiáng)鐵死亡、抑制腫瘤生長(zhǎng)。低表達(dá)AAK1與乳腺癌不良預(yù)后相關(guān)。研究揭示“外源鐵攝取”是鐵死亡關(guān)鍵步驟,提出靶向PKCβⅡ-AAK1-AP2M1軸促進(jìn)鐵死亡的抗腫瘤新策略。
03、靶點(diǎn):GLUD1
應(yīng)用:腫瘤免疫治療的潛在治療靶點(diǎn)
來源:Tumor-produced ammonia is metabolized by regulatory T cells to further impede anti-tumor immunity.Cell,2026 Jan 22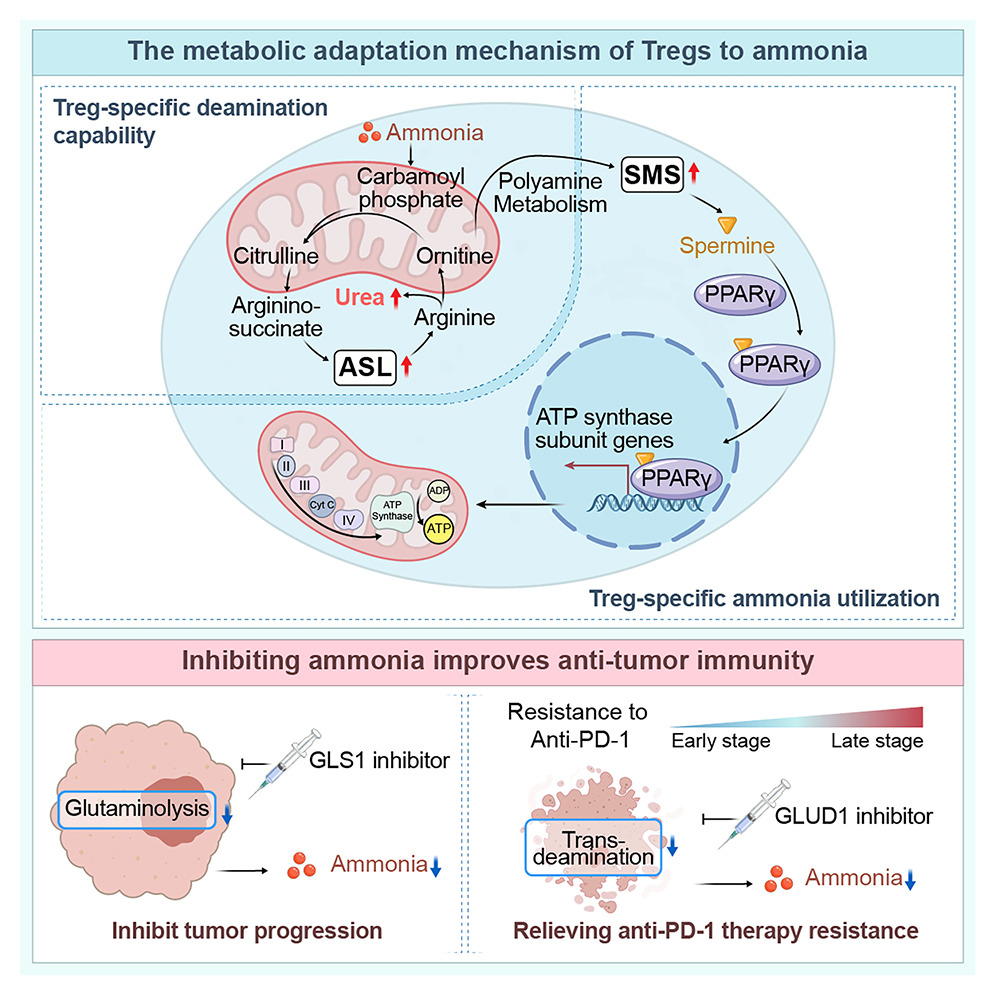
徐州醫(yī)科大學(xué)呂凌團(tuán)隊(duì)12月《Cell》報(bào)道,腫瘤谷氨酰胺代謝釋放的氨在局部形成高濃度免疫抑制微區(qū),Treg借GLUD1-尿素循環(huán)/多胺軸將氨轉(zhuǎn)化為生存與功能優(yōu)勢(shì):ASL解毒、FOXP3-SMS-精胺-PPARγ增強(qiáng)氧化磷酸化,實(shí)現(xiàn)富集并抑制CD8+T細(xì)胞。抗PD-1治療死亡細(xì)胞進(jìn)一步釋放氨加劇Treg強(qiáng)化。靶向GLUD1降低氨水平可削弱Treg、協(xié)同免疫檢查點(diǎn)抑制劑,為克服耐藥提供新靶點(diǎn)。
04、靶點(diǎn):VWF
應(yīng)用:特發(fā)性肺纖維化的治療
來源:von Willebrand Factor Deficiency Inhibits Endothelial-to-Mesenchymal Transition to Attenuate Pulmonary Fibrosis.Am J Respir Cell Mol Biol,2025 Dec
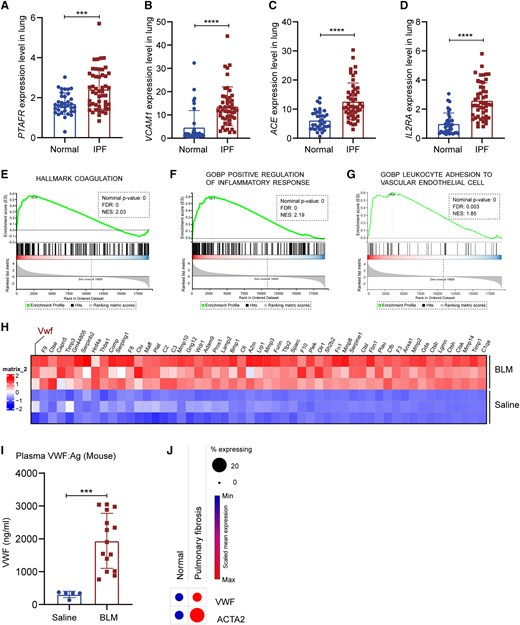
上海藥物所宮麗崑/復(fù)旦大學(xué)朱棣/上海交大醫(yī)學(xué)院王炳順聯(lián)合團(tuán)隊(duì)于AJRCMB首次揭示,內(nèi)皮損傷標(biāo)志物血管性血友病因子(VWF)是特發(fā)性肺纖維化(IPF)進(jìn)展的關(guān)鍵驅(qū)動(dòng)。公共數(shù)據(jù)、人肺微血管內(nèi)皮細(xì)胞及博來霉素小鼠模型一致顯示,VWF缺失/敲低通過阻斷Wnt/β-catenin信號(hào),抑制內(nèi)皮-間質(zhì)轉(zhuǎn)化(EndoMT),維持血管內(nèi)皮屏障,減少巨噬浸潤(rùn),從而顯著減輕膠原沉積并提高存活率。研究提出靶向VWF可逆轉(zhuǎn)現(xiàn)行藥物無法遏制的肺纖維化,為IPF抗纖維化治療提供新靶點(diǎn)。
05、靶點(diǎn):SVEP1
應(yīng)用:肝內(nèi)膽管癌(特別是SVEP1低表達(dá)型)的潛在治療靶點(diǎn)
來源:Low SVEP1 in intrahepatic cholangiocarcinoma mediates phenotype switching-driven metastasis by Jag2/Notch1/Hes5.Cell Death Dis,2025 Nov 28
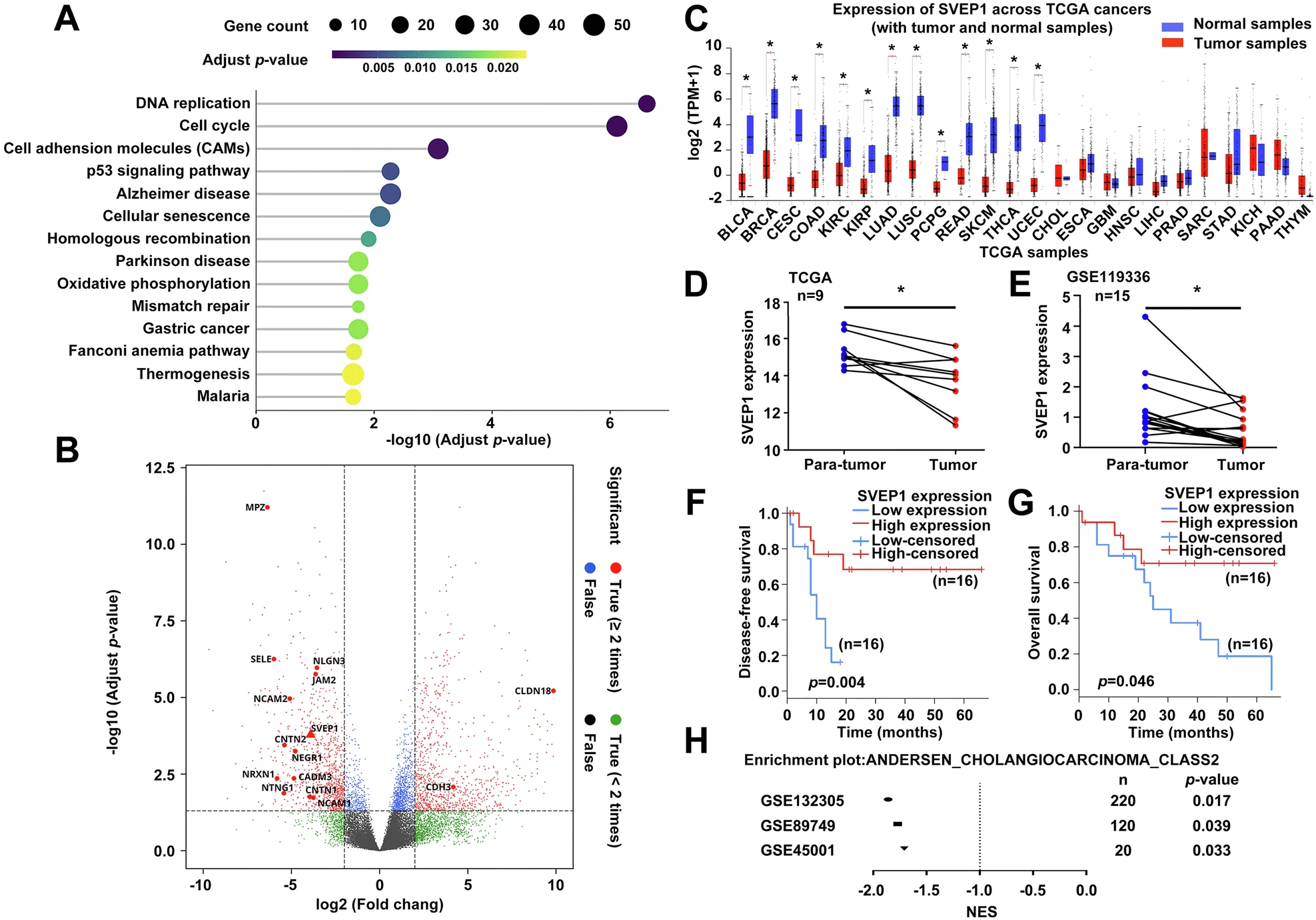
Cell Death Dis研究首次報(bào)道ECM蛋白SVEP1在肝內(nèi)膽管癌(ICC)中顯著低表達(dá),是獨(dú)立不良預(yù)后標(biāo)志。機(jī)制上,SVEP1缺失解除對(duì)Notch配體Jag2的膜上“物理束縛”,激活Jag2/Notch1/Hes5軸,驅(qū)動(dòng)EMT樣表型轉(zhuǎn)換,增強(qiáng)遷移、侵襲與肺轉(zhuǎn)移;阻斷該軸可逆轉(zhuǎn)惡性表型。體內(nèi)外實(shí)驗(yàn)證實(shí)SVEP1為轉(zhuǎn)移抑制因子,靶向Jag2/Notch1通路或可為SVEP1低表達(dá)ICC患者提供新治療策略。
06、靶點(diǎn):G6PC1
應(yīng)用:肝細(xì)胞癌(低G6PC1型)的潛在治療靶點(diǎn)
來源:G6PC1 expression as a prognostic biomarker associated with metabolic reprogramming and tumor microenvironment in hepatocellular carcinoma.Front Immunol,2025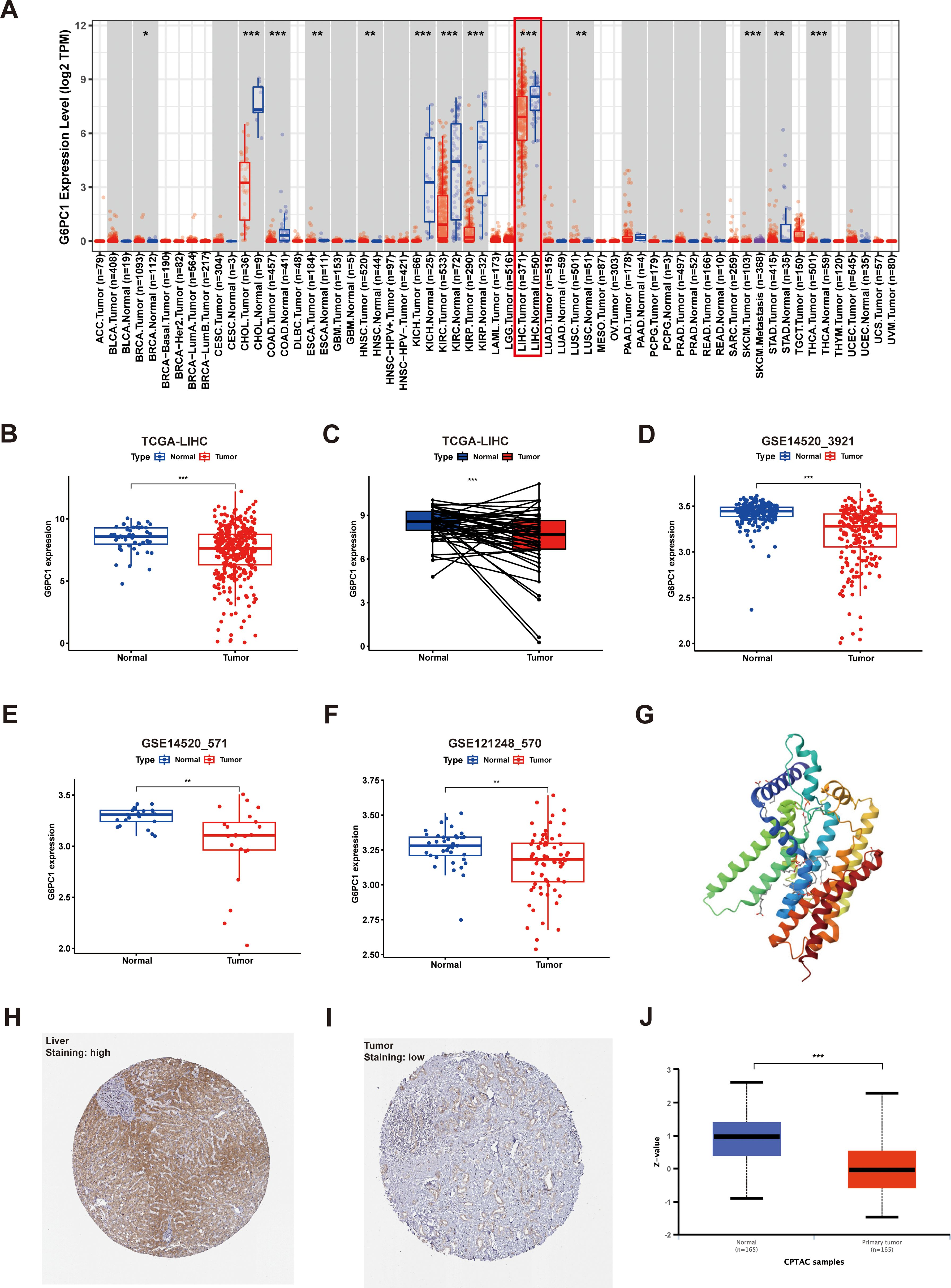
Front Immunol研究基于TCGA等數(shù)據(jù)庫(kù)發(fā)現(xiàn)糖異生限速酶G6PC1在HCC組織中顯著下調(diào),是獨(dú)立不良預(yù)后因子。低表達(dá)通過上調(diào)PKM、G6PD強(qiáng)化糖酵解,并招募M2巨噬細(xì)胞、Treg等塑造免疫抑制微環(huán)境,降低索拉非尼敏感性;但其高M(jìn)SI/TMB預(yù)示抗PD-L1療效更佳。體外過表達(dá)G6PC1可抑制PKM/G6PD并減弱增殖,提示靶向代謝酶或聯(lián)合免疫治療可為低G6PC1高危患者提供個(gè)體化策略。
07、靶點(diǎn):CYSLTR2
應(yīng)用:CysLT2R相關(guān)炎癥及癌癥相關(guān)的潛在治療靶點(diǎn)
來源:Molecular insights into ago-allosteric modulation at Cysteinyl leukotriene receptor 2.Nat Commun,2025 Dec 16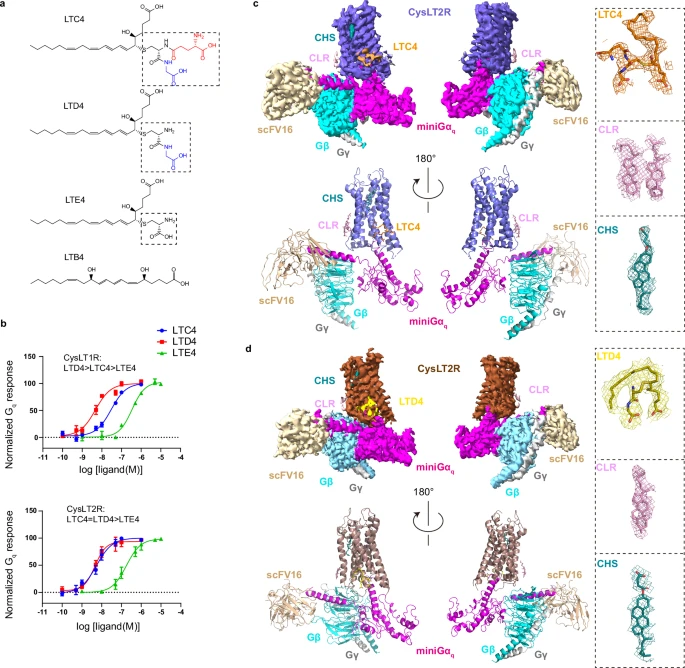
Nat Commun報(bào)道,廣州醫(yī)科大學(xué)劉恒團(tuán)隊(duì)與中科院深圳先進(jìn)院王崇元團(tuán)隊(duì)解析人源CysLT2R與內(nèi)源配體LTC4/LTD4及Gq蛋白的冷凍電鏡結(jié)構(gòu),發(fā)現(xiàn)半胱氨酰白三烯并非結(jié)合傳統(tǒng)胞外正構(gòu)口袋,而是嵌入由TM3-5/ICL2圍成的胞內(nèi)側(cè)別構(gòu)位,充當(dāng)“別構(gòu)-正向協(xié)同激動(dòng)劑”,直接抬升ICL2并觸發(fā)TM6外移完成Gq招募;該機(jī)制可協(xié)同神經(jīng)酰胺共同激活受體,為設(shè)計(jì)別構(gòu)或雙位點(diǎn)調(diào)節(jié)劑、實(shí)現(xiàn)精準(zhǔn)抑制CysLT2R相關(guān)炎癥及癌癥提供全新結(jié)構(gòu)模板。
08、靶點(diǎn):NRF1
應(yīng)用:炎性衰老及相關(guān)疾病的潛在治療靶點(diǎn)
來源:NRF1-mediated innate immune response drives inflammaging.Nat Commun,2025 Dec 11
Nat Commun報(bào)道,南開大學(xué)朱玉山、李艷君團(tuán)隊(duì)發(fā)現(xiàn)核呼吸因子1(NRF1)通過直接轉(zhuǎn)錄激活固有免疫關(guān)鍵因子TBK1和IRF3,驅(qū)動(dòng)“ATM–NRF1–TBK1/IRF3–Ⅰ型干擾素”正反饋通路,從而加劇衰老相關(guān)分泌表型(SASP)并促進(jìn)炎性衰老;在老年小鼠中敲低NRF1可有效抑制多器官炎癥、延緩衰老表型,揭示了NRF1是連接DNA損傷、固有免疫與炎性衰老的關(guān)鍵轉(zhuǎn)錄樞紐,為開發(fā)靶向炎性衰老及相關(guān)疾病的藥物提供了新理論依據(jù)與潛在靶點(diǎn)。
09、靶點(diǎn):ALDH1B1
應(yīng)用:肺癌轉(zhuǎn)移的潛在治療靶點(diǎn)
來源:Compression-induced NF-κB activation sustains tumor cell survival in confinement by detoxifying aldehydes and promotes metastasis.Nat Commun,2025 Dec 14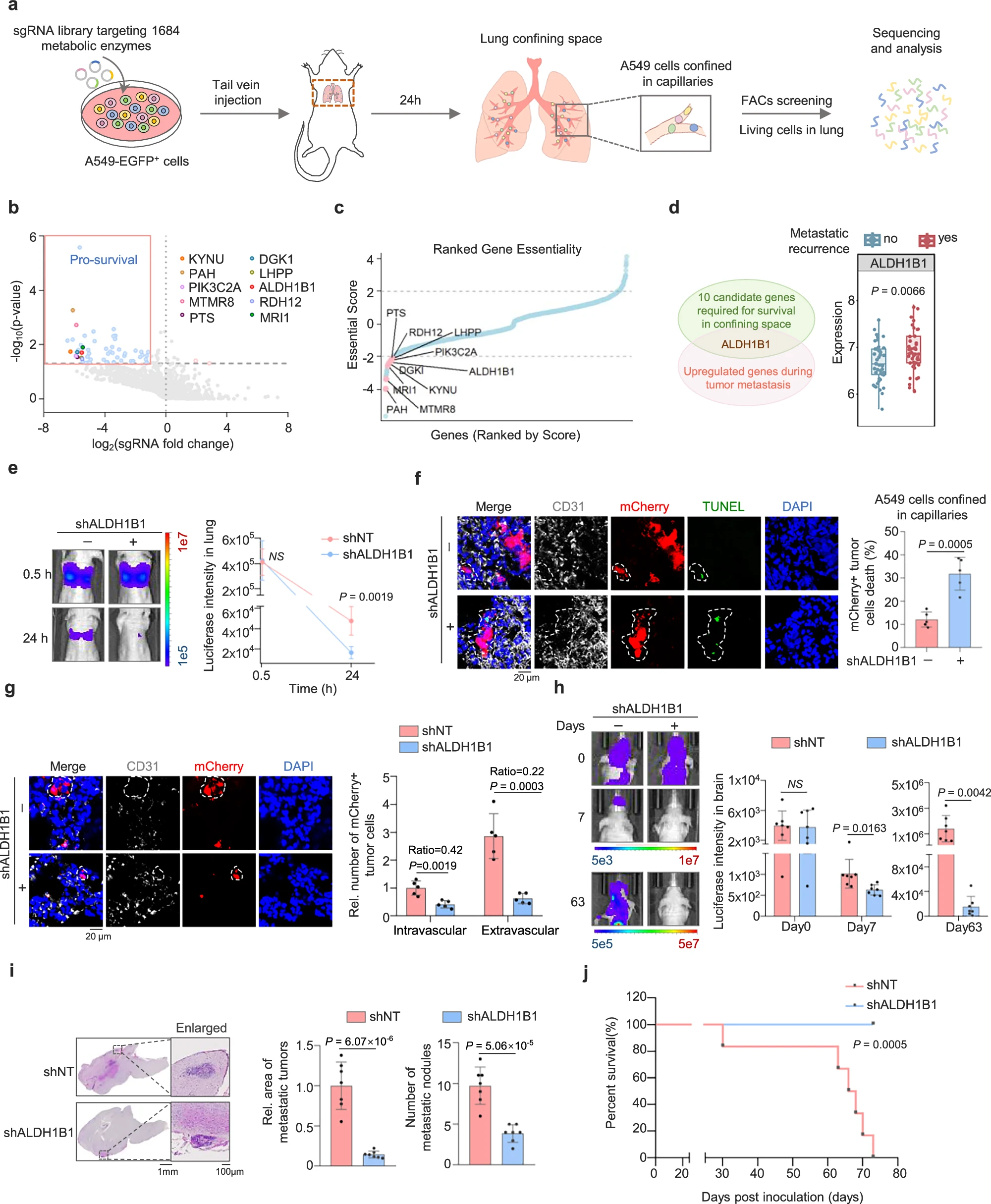
10、靶點(diǎn):LRBA
應(yīng)用:腫瘤免疫治療的潛在治療靶點(diǎn)
來源:Targeting LRBA triggers CTLA4 degradation and antitumor immunity for cancer immunotherapy.Nat Commun,2025 Dec 14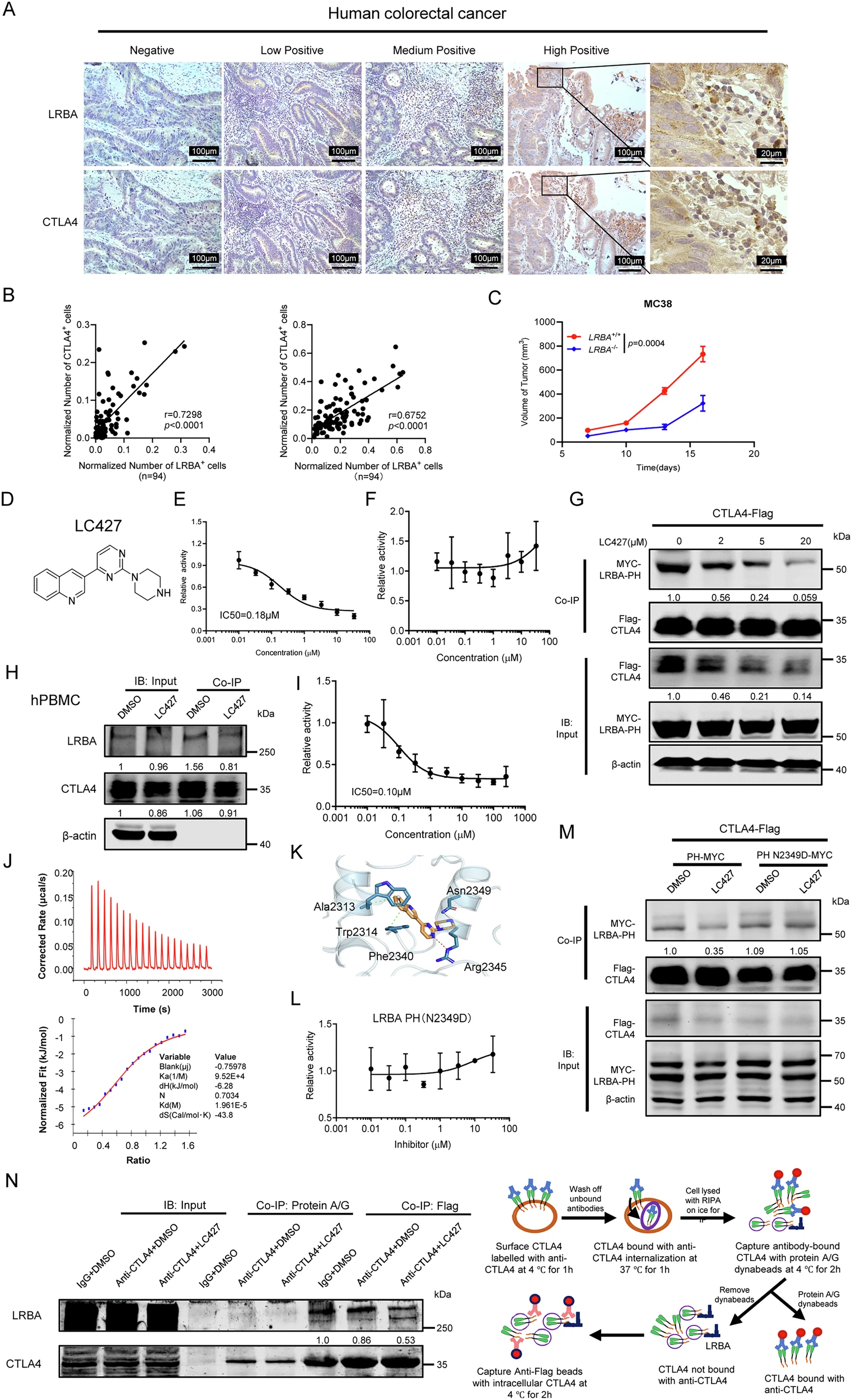
推薦產(chǎn)品
參考文獻(xiàn)
[1] METTL14 integrates tumor-derived SAM to drive parabrachial epigenetic rewiring in pancreatic cancer.Neuron,2026 Jan 21
應(yīng)用:PDAC疼痛抑郁共病的潛在治療靶點(diǎn)
來源:METTL14 integrates tumor-derived SAM to drive parabrachial epigenetic rewiring in pancreatic cancer.Neuron,2026 Jan 21

圖源:10.1016/j.neuron.2025.10.002[1]
中山大學(xué)謝敬敦團(tuán)隊(duì)11月于《Neuron》報(bào)道,PDAC小鼠血漿及外側(cè)臂旁核(LPBN)內(nèi)腫瘤源S-腺苷甲硫氨酸(SAM)升高,激活谷氨酸能神經(jīng)元METTL14,經(jīng)m6A修飾增強(qiáng)ADM mRNA穩(wěn)定性,使神經(jīng)元超興奮,并通過LPBNGlu→PVTGlu/LHGlu環(huán)路誘發(fā)痛覺過敏與抑郁。敲低METTL14、抑制SAM合成或低甲硫氨酸飲食均可逆轉(zhuǎn)共病并抑制腫瘤,揭示“腫瘤-代謝物-神經(jīng)元”信號(hào)軸,為PDAC疼痛抑郁共病提供可轉(zhuǎn)化干預(yù)策略。
02、靶點(diǎn):AAK1
應(yīng)用:乳腺癌等實(shí)體瘤的潛在治療靶點(diǎn)
來源:AAK1 activation-mediated iron trafficking drives ferroptotic cell death.Nat Commun,2025 Dec 17

圖源:10.1038/s41467-025-67523-9[2]
中山大學(xué)朱孝峰團(tuán)隊(duì)12月在Nat Commun報(bào)道,鐵死亡啟動(dòng)時(shí)PKCβII感應(yīng)脂質(zhì)過氧化,并在S670/T674位點(diǎn)磷酸化激活A(yù)AK1,后者磷酸化AP2M1促進(jìn)網(wǎng)格蛋白依賴的轉(zhuǎn)鐵蛋白受體TFR1內(nèi)吞,使胞外鐵大量流入,為脂質(zhì)過氧化物生成提供催化鐵,從而驅(qū)動(dòng)腫瘤細(xì)胞鐵死亡。敲除或抑制PKCβⅡ-AAK1-AP2M1軸均降低鐵攝取、抑制鐵死亡并削弱放療敏感性;激活該軸則增強(qiáng)鐵死亡、抑制腫瘤生長(zhǎng)。低表達(dá)AAK1與乳腺癌不良預(yù)后相關(guān)。研究揭示“外源鐵攝取”是鐵死亡關(guān)鍵步驟,提出靶向PKCβⅡ-AAK1-AP2M1軸促進(jìn)鐵死亡的抗腫瘤新策略。
03、靶點(diǎn):GLUD1
應(yīng)用:腫瘤免疫治療的潛在治療靶點(diǎn)
來源:Tumor-produced ammonia is metabolized by regulatory T cells to further impede anti-tumor immunity.Cell,2026 Jan 22

圖源:10.1016/j.cell.2025.11.034[3]
徐州醫(yī)科大學(xué)呂凌團(tuán)隊(duì)12月《Cell》報(bào)道,腫瘤谷氨酰胺代謝釋放的氨在局部形成高濃度免疫抑制微區(qū),Treg借GLUD1-尿素循環(huán)/多胺軸將氨轉(zhuǎn)化為生存與功能優(yōu)勢(shì):ASL解毒、FOXP3-SMS-精胺-PPARγ增強(qiáng)氧化磷酸化,實(shí)現(xiàn)富集并抑制CD8+T細(xì)胞。抗PD-1治療死亡細(xì)胞進(jìn)一步釋放氨加劇Treg強(qiáng)化。靶向GLUD1降低氨水平可削弱Treg、協(xié)同免疫檢查點(diǎn)抑制劑,為克服耐藥提供新靶點(diǎn)。
04、靶點(diǎn):VWF
應(yīng)用:特發(fā)性肺纖維化的治療
來源:von Willebrand Factor Deficiency Inhibits Endothelial-to-Mesenchymal Transition to Attenuate Pulmonary Fibrosis.Am J Respir Cell Mol Biol,2025 Dec

圖源:10.1165/rcmb.2024-0527OC[4]
上海藥物所宮麗崑/復(fù)旦大學(xué)朱棣/上海交大醫(yī)學(xué)院王炳順聯(lián)合團(tuán)隊(duì)于AJRCMB首次揭示,內(nèi)皮損傷標(biāo)志物血管性血友病因子(VWF)是特發(fā)性肺纖維化(IPF)進(jìn)展的關(guān)鍵驅(qū)動(dòng)。公共數(shù)據(jù)、人肺微血管內(nèi)皮細(xì)胞及博來霉素小鼠模型一致顯示,VWF缺失/敲低通過阻斷Wnt/β-catenin信號(hào),抑制內(nèi)皮-間質(zhì)轉(zhuǎn)化(EndoMT),維持血管內(nèi)皮屏障,減少巨噬浸潤(rùn),從而顯著減輕膠原沉積并提高存活率。研究提出靶向VWF可逆轉(zhuǎn)現(xiàn)行藥物無法遏制的肺纖維化,為IPF抗纖維化治療提供新靶點(diǎn)。
05、靶點(diǎn):SVEP1
應(yīng)用:肝內(nèi)膽管癌(特別是SVEP1低表達(dá)型)的潛在治療靶點(diǎn)
來源:Low SVEP1 in intrahepatic cholangiocarcinoma mediates phenotype switching-driven metastasis by Jag2/Notch1/Hes5.Cell Death Dis,2025 Nov 28

圖源:10.1038/s41419-025-08170-2[5]
Cell Death Dis研究首次報(bào)道ECM蛋白SVEP1在肝內(nèi)膽管癌(ICC)中顯著低表達(dá),是獨(dú)立不良預(yù)后標(biāo)志。機(jī)制上,SVEP1缺失解除對(duì)Notch配體Jag2的膜上“物理束縛”,激活Jag2/Notch1/Hes5軸,驅(qū)動(dòng)EMT樣表型轉(zhuǎn)換,增強(qiáng)遷移、侵襲與肺轉(zhuǎn)移;阻斷該軸可逆轉(zhuǎn)惡性表型。體內(nèi)外實(shí)驗(yàn)證實(shí)SVEP1為轉(zhuǎn)移抑制因子,靶向Jag2/Notch1通路或可為SVEP1低表達(dá)ICC患者提供新治療策略。
06、靶點(diǎn):G6PC1
應(yīng)用:肝細(xì)胞癌(低G6PC1型)的潛在治療靶點(diǎn)
來源:G6PC1 expression as a prognostic biomarker associated with metabolic reprogramming and tumor microenvironment in hepatocellular carcinoma.Front Immunol,2025

圖源:10.3389/fimmu.2025.1623315[6]
Front Immunol研究基于TCGA等數(shù)據(jù)庫(kù)發(fā)現(xiàn)糖異生限速酶G6PC1在HCC組織中顯著下調(diào),是獨(dú)立不良預(yù)后因子。低表達(dá)通過上調(diào)PKM、G6PD強(qiáng)化糖酵解,并招募M2巨噬細(xì)胞、Treg等塑造免疫抑制微環(huán)境,降低索拉非尼敏感性;但其高M(jìn)SI/TMB預(yù)示抗PD-L1療效更佳。體外過表達(dá)G6PC1可抑制PKM/G6PD并減弱增殖,提示靶向代謝酶或聯(lián)合免疫治療可為低G6PC1高危患者提供個(gè)體化策略。
07、靶點(diǎn):CYSLTR2
應(yīng)用:CysLT2R相關(guān)炎癥及癌癥相關(guān)的潛在治療靶點(diǎn)
來源:Molecular insights into ago-allosteric modulation at Cysteinyl leukotriene receptor 2.Nat Commun,2025 Dec 16

圖源:10.1038/s41467-025-67630-7[7]
Nat Commun報(bào)道,廣州醫(yī)科大學(xué)劉恒團(tuán)隊(duì)與中科院深圳先進(jìn)院王崇元團(tuán)隊(duì)解析人源CysLT2R與內(nèi)源配體LTC4/LTD4及Gq蛋白的冷凍電鏡結(jié)構(gòu),發(fā)現(xiàn)半胱氨酰白三烯并非結(jié)合傳統(tǒng)胞外正構(gòu)口袋,而是嵌入由TM3-5/ICL2圍成的胞內(nèi)側(cè)別構(gòu)位,充當(dāng)“別構(gòu)-正向協(xié)同激動(dòng)劑”,直接抬升ICL2并觸發(fā)TM6外移完成Gq招募;該機(jī)制可協(xié)同神經(jīng)酰胺共同激活受體,為設(shè)計(jì)別構(gòu)或雙位點(diǎn)調(diào)節(jié)劑、實(shí)現(xiàn)精準(zhǔn)抑制CysLT2R相關(guān)炎癥及癌癥提供全新結(jié)構(gòu)模板。
08、靶點(diǎn):NRF1
應(yīng)用:炎性衰老及相關(guān)疾病的潛在治療靶點(diǎn)
來源:NRF1-mediated innate immune response drives inflammaging.Nat Commun,2025 Dec 11

圖源:10.1038/s41467-025-66368-6[8]
Nat Commun報(bào)道,南開大學(xué)朱玉山、李艷君團(tuán)隊(duì)發(fā)現(xiàn)核呼吸因子1(NRF1)通過直接轉(zhuǎn)錄激活固有免疫關(guān)鍵因子TBK1和IRF3,驅(qū)動(dòng)“ATM–NRF1–TBK1/IRF3–Ⅰ型干擾素”正反饋通路,從而加劇衰老相關(guān)分泌表型(SASP)并促進(jìn)炎性衰老;在老年小鼠中敲低NRF1可有效抑制多器官炎癥、延緩衰老表型,揭示了NRF1是連接DNA損傷、固有免疫與炎性衰老的關(guān)鍵轉(zhuǎn)錄樞紐,為開發(fā)靶向炎性衰老及相關(guān)疾病的藥物提供了新理論依據(jù)與潛在靶點(diǎn)。
09、靶點(diǎn):ALDH1B1
應(yīng)用:肺癌轉(zhuǎn)移的潛在治療靶點(diǎn)
來源:Compression-induced NF-κB activation sustains tumor cell survival in confinement by detoxifying aldehydes and promotes metastasis.Nat Commun,2025 Dec 14

圖源:10.1038/s41467-025-67452-7[9]
Nature Commun發(fā)表中科院分子細(xì)胞卓越中心楊巍維組與交大胸科姚烽、新華醫(yī)院陶幫寶合作成果:利用CRISPR體內(nèi)篩選發(fā)現(xiàn)醛脫氫酶ALDH1B1是腫瘤細(xì)胞穿越狹窄血管時(shí)的存活關(guān)鍵。機(jī)械擠壓力促使CSK23結(jié)合并磷酸化IKKβ,激活NF-κB上調(diào)ALDH1B1,增強(qiáng)醛解毒并抑制鐵死亡,助力小鼠肺癌遠(yuǎn)端轉(zhuǎn)移。研究首次闡明“機(jī)械力-NF-κB-ALDH1B1-醛代謝”耦合軸,為靶向CSK23或ALDH1B1阻斷腫瘤轉(zhuǎn)移提供新策略。10、靶點(diǎn):LRBA
應(yīng)用:腫瘤免疫治療的潛在治療靶點(diǎn)
來源:Targeting LRBA triggers CTLA4 degradation and antitumor immunity for cancer immunotherapy.Nat Commun,2025 Dec 14

圖源:10.1038/s41467-025-67365-5[10]
Nat Commun報(bào)道,廣州中醫(yī)藥大學(xué)-上海交大團(tuán)隊(duì)揭示囊泡運(yùn)輸?shù)鞍譒RBA通過穩(wěn)定CTLA4負(fù)調(diào)免疫,首創(chuàng)口服小分子LC427阻斷LRBA–CTLA4互作,加速CTLA4降解。小鼠模型中,LC427單藥抑制腫瘤生長(zhǎng)并增CD8?T浸潤(rùn),與PD-1抗體協(xié)同克服耐藥;在結(jié)腸炎模型幾乎不誘發(fā)irAEs,實(shí)現(xiàn)療效-毒性解耦。研究提出“細(xì)胞內(nèi)穩(wěn)態(tài)精準(zhǔn)調(diào)控”新范式,為高危免疫檢查點(diǎn)藥物開發(fā)提供通用篩選平臺(tái)。推薦產(chǎn)品
| 靶點(diǎn) | 重組蛋白 | 貨號(hào) |
| AAK1 | Recombinant Human AP2-associated protein kinase 1 (AAK1), partial | CSB-MP643571HU |
| ALDH1B1 | Recombinant Human Aldehyde dehydrogenase X, mitochondrial (ALDH1B1), partial | CSB-EP001568HU1 |
| CYSLTR2 | Recombinant Human Cysteinyl leukotriene receptor 2 (CYSLTR2), partial | CSB-MP006466HU1 |
| G6PC1 | Recombinant Human Glucose-6-phosphatase (G6PC), partial | CSB-EP009118HU1a2 |
| GLUD1 | Recombinant Human Glutamate dehydrogenase 1, mitochondrial (GLUD1) | CSB-MP009544HU |
| LRBA | Recombinant Human Lipopolysaccharide-responsive and beige-like anchor protein (LRBA), partial | CSB-EP013070HU(C) |
| METTL14 | Recombinant Human N6-adenosine-methyltransferase non-catalytic subunit (METTL14) | CSB-EP884519HU |
| NRF1 | Recombinant Mouse Nuclear respiratory factor 1 (Nrf1) | CSB-EP016076MO |
| SVEP1 | Recombinant Human Sushi, von Willebrand factor type A, EGF and pentraxin domain-containing protein 1 (SVEP1), partial | CSB-MP689809HU |
| VWF | Recombinant Mouse von Willebrand factor (Vwf), partial | CSB-EP025960MO |
參考文獻(xiàn)
[1] METTL14 integrates tumor-derived SAM to drive parabrachial epigenetic rewiring in pancreatic cancer.Neuron,2026 Jan 21
[2]AAK1 activation-mediated iron trafficking drives ferroptotic cell death.Nat Commun,2025 Dec 17
[3]Tumor-produced ammonia is metabolized by regulatory T cells to further impede anti-tumor immunity.Cell,2026 Jan 22
[4]von Willebrand Factor Deficiency Inhibits Endothelial-to-Mesenchymal Transition to Attenuate Pulmonary Fibrosis.Am J Respir Cell Mol Biol,2025 Dec
[5]Low SVEP1 in intrahepatic cholangiocarcinoma mediates phenotype switching-driven metastasis by Jag2/Notch1/Hes5.Cell Death Dis,2025 Nov 28
[6]G6PC1 expression as a prognostic biomarker associated with metabolic reprogramming and tumor microenvironment in hepatocellular carcinoma.Front Immunol,2025
[7]Molecular insights into ago-allosteric modulation at Cysteinyl leukotriene receptor 2.Nat Commun,2025 Dec 16
[8]NRF1-mediated innate immune response drives inflammaging.Nat Commun,2025 Dec 11
[9]Compression-induced NF-κB activation sustains tumor cell survival in confinement by detoxifying aldehydes and promotes metastasis.Nat Commun,2025 Dec 14
[10]Targeting LRBA triggers CTLA4 degradation and antitumor immunity for cancer immunotherapy.Nat Commun,2025 Dec 14
*免責(zé)聲明:華美生物內(nèi)容團(tuán)隊(duì)僅是分享和解讀公開研究論文及其發(fā)現(xiàn),本文僅作信息交流,文中觀點(diǎn)不代表華美生物立場(chǎng),請(qǐng)理解。











